Contact Us
Latest News
- Clues beginning to emerge on asymtomatic SARS-CoV-2 infection
- Back in November of 2020, during the first wave of the COVID-19 pandemic, I was teaching an in-person microbiology laboratory. One of my students had just been home to see his parents, and they all c…
- Read more
- Could there maybe be better uses of genetics and probiotics?
- Professor Meng Dong and his laboratory have created a probiotic that can metabolize alcohol quickly and maybe prevent some of the adverse effects of alcohol consumption. The scientists cloned a highl…
- Read more
- ChatGPT is not the end of essays in education
- The takeover of AI is upon us! AI can now take all our jobs, is the click-bait premise you hear from the news. While I cannot predict the future, I am dubious that AI will play such a dubious role in…
- Read more
- Fighting infections with infections
- Multi-drug-resistant bacterial infections are becoming more of an issue, with 1.2 million people dying of previously treatable bacterial infections. Scientists are frantically searching for new metho…
- Read more
- A tale of two colleges
- COVID-19 at the University of Wisconsin this fall has been pretty much a non-issue. While we are wearing masks, full in-person teaching is happening on campus. Bars, restaurants, and all other busine…
- Read more
( 34110 Reads)
None Max
Learning Objectives
After reading this section, students will be able to...
- Be able to read and write the chemical and structure formula of important molecules.
- Be able to recognize common carbon groups found in biological molecules.
- Describe the properties of water that make it so crucial for biological life.
- Define pH and identify its importance in living systems.
- Explain the unique properties of carbon and how these make it central in biological structures.
A common terminology for representing atoms and compounds has been developed to help communicate their composition and the forces that hold them together. The most important for our purpose are the chemical formula and the structural formula. The chemical formula is a type of shorthand that lists the makeup of any compound. Table 2.2 lists the symbols used for the common elements in biological molecules. A chemical formula contains the symbol for each constituent atom and its ratio in the compound. If more than one atom of the same type is present in a molecule, a subscript following the symbol designates the number (Table 2.3). The structural formula shows the arrangement of atoms in the compound. Each atom of the molecule is drawn using its symbol, and lines are drawn between them, indicating the covalent bonds formed.
Table 2.3. Comparison of Chemical Formula, Structural Formula, and Molecular Weight.
| Compound | Chemical Formula | Structural Formula§ | Molecular Weight* | Comments |
| Hydrogen | H2 | 1 | 2.01 | |
| Methane | CH4 | 2 | 16.04 | The major component of natural gas |
| Ethanol | C2 H6 O | 3 | 46.06 | Common alcohol found in beer, wine and liquors |
| Acetic acid | C2 H4 O2 | 4 | 60.05 | Major component of vinegar |
| Glucose | C6 H1 2 O6 | 5 | 180.16 | Common sugar in candies and soft drinks |
| Sucrose | C1 2 H2 2 O1 1 | 6 | 342.30 | Table sugar |
| Oleic Acid | C1 8 H2 4 O2 | 7 | 282.47 | Major fatty acid in butter |
§ Structural formulas are shown in Figure 2.6
*Molecular weights are never exact integers dues to the existence of isotopes for each element. The atomic weight of each atom is an average of the isotopes commonly found.
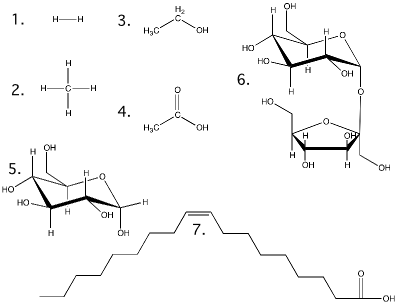
Figure 2.6. Structural formulas for the molecules in Table 2.3. Show here are the structures for the molecules discussed in table 2.3
In biological chemistry, much of the structure of a molecule is made up of carbon and hydrogen atoms. Carbon will form the molecule's backbone, and hydrogen will take up extra bonds that do not link to any other atom. Organic chemists have developed a further shorthand of not writing in the hydrogen atoms and leaving out the carbon symbol. This shorthand greatly reduces the work of drawing structures and increases the structural clarity of the molecule. We will follow this convention throughout most of this book.
It is also important to understand the procedure for measuring and weighing atoms and molecules. The weight of an atom is the sum of the protons and neutrons it contains in its nucleus. The molecular weight is the sum of the weights of all the atoms that make up the molecule. Since it is difficult to work with one molecule of anything, scientists created the term gram molecular weight. The gram molecular weight is the weight of 6.022 x 1023 molecules of a compound. For each molecule, this works out to exactly the molecular weight of the compound in grams. For example, the molecular weight of water is 18 Daltons (1 oxygen : 16 Daltons; 2 hydrogen: 2 Daltons), and the gram molecular weight is 18 grams. A mole of a compound is defined as one gram molecular weight. Finally, the concentration of a compound dissolved in water is most conveniently expressed as molarity. Scientists define this as the number of moles of a compound per liter of water. Figure 2.7 shows a summary of these conversions.

Figure 2.7. Conversion of grams to moles to molarity.. Sodium Chloride has a molecular weight of 58.44, which is the weight of 6.022 x 1023 molecules. If 58.44 grams are weight out, that would be one mole of NaCl. If this is then dissolved in water it would be a 1 molar solution of NaCl.
Water
Water is the most common molecule on the earth's outer layer, covering 75% of the earth's surface. Because of its unique properties, it is the universal solvent for the chemistry of life. You are more than 60% water and would die if your supply were cut off for more than a few days. The oxygen atoms in water pull the electrons toward themselves, causing the hydrogen atoms to be electron starved. The hydrogen in water will seek to form hydrogen bonds with the oxygen of nearby water molecules. This stickiness is termed polarity. Water is not the only polar molecule, any molecule where the electrons are not shared equally will be polar, but water is one of the smallest and most common polar molecules. Because of its polarity and size, water has several unique properties that make it so essential to living systems:
- Water is an excellent solvent. More things dissolve in water than any other solvent. Because of its polar nature, water can cause ionic compounds, like salt, to dissociate into their constituent atoms freeing them to participate in chemical reactions. Enzymes and many of the biomolecules of living systems are soluble in water.
- Water sticks to things, which results in capillarity, the ability to move through small orifices or fine pores against the force of gravity and other forces. This tendency is crucial for moving water about in plants, animals, and microorganisms.
- Water has a high specific heat, meaning that it requires a large amount of heat to change its temperature. Since organisms are mostly water, they lose and gain heat slowly, providing a stable or slowly changing temperature.
- Because of its polarity, water has a high boiling point allowing life to exist in a large range of temperatures.
pH profoundly affects biological activities
The cell cytoplasm is an aqueous environment and acids can dissociate into hydrogen ions (H+) and anions. The higher the H+ concentration, the more acidic the solution. Bases in solution can also dissociate into OH- ions and cations. A more basic solution will have a higher concentration of OH-. This balance in a solution can be quantified by measuring the hydrogen ion concentration, which can vary over a wide range. The concept of pH (potential of hydrogen) was developed as a kind of shorthand for the concentration of hydrogen ions and is defined in the following equation
pH = -log[H+]
Where H+ is the concentration of hydrogen ions in moles per liter.
In normal aqueous solutions, pH values will range from 0 to 14. A pH of 14 indicates a hydrogen ion concentration of 1 x 10-14 moles per liter. A pH of 0 indicates a hydrogen ion concentration of 1 mole per liter, a very acidic solution! Human body fluids have a pH of about 7 or a neutral pH where the concentration of hydrogen ions equals the concentration of hydroxide ions. Acidic solutions contain more H+ than OH- and have a pH below 7, while basic solutions have more OH- than H+ and have a pH above 7. Figure 2.8 shows the pH scale and the pH of some fluids you are probably familiar with.
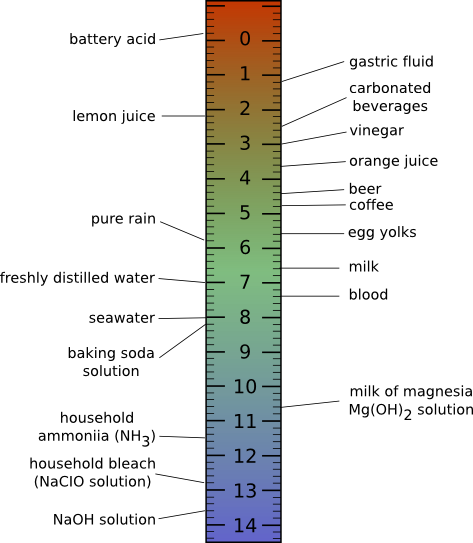
Figure 2.8. The pH scale and the pH of some common foods.. At low pH, the hydrogen ion concentration is high, as the pH increases, the concentration of hydrogen ions decrease and the concentration of hydroxide ions increase. Acidic solutions have more hydrogen ions and basic solutions have more hydroxide ions. The pH of common items is also shown
Maintaining the pH of the cytoplasm is critical to cell function. Small changes in pH can have profound effects on the cell. Many enzymatic reactions require the use of H+ or OH- and changing the pH of the cytoplasm can cause them to no longer function. In addition, enzyme stability can be affected by pH, causing enzymes to denature. Larger changes can even cause the cells chromosome to destabilize.
The cells collection of acids and bases is constantly changing as it moves through its environment, takes up nutrients, carries out metabolism, and excretes waste products. Cells maintain their pH using molecules that can serve as buffers. Buffers are molecular sponges that soak up H+ or OH-. These molecules readily react with H+ and OH- and prevent drastic changes to the pH of the cytoplasm. Microbes that grow in extreme environments of very high or low pH will even have pumps that pump H+ and/or OH- in or out of the cell. The goal is always to maintain the cytoplasm at a near-neutral pH. In low pH environments, cell pump H+ out, while in high pH environments, cells cell pump H+ in.
In contrast to the cytoplasm of the cell, the outside environment can have a wide range of pH, which can dictate what types of microbes can thrive. Soils and water, often near-neutral pH, can become acidic or basic depending upon the mix of minerals, pollutants, and the action of microbes themselves. Thiobacillus ferroxidans can grow in high sulfur coal piles, converting the sulfur to sulfuring acid. If industries leave coal piles in the open air, water flowing through them can become rivers of sulfuric acid, as low as pH 2. This microbe will happily grow under these conditions. Microbes growing in a laboratory medium, or in milk, can produce acidic end products that drop the pH. This result is rarely desirable in the laboratory, and buffers are often added to media to prevent pH changes. However, the growth of microbes in milk, and it's acidification, is often desirable in the formation of delectable treats such as yogurt, buttermilk, and cheese. No matter the pH of the environment, there are almost always microbes capable of growing under these conditions.
Carbon is particularly central to biology
Carbon can rearrange its 1s and 3 p orbitals in its outermost unfilled shell into four equal orbitals that project out in a pyramid-like organization. Each orbital contains one electron, forming four equal bonds instead of the two one would typically expect. This unique property enables it to bond with other atoms to form intricate lattices of infinite variation. Organic molecules (as carbon-containing compounds are called) use carbon backbones as the skeleton for building all sorts of structures that can perform seemingly limitless functions. If carbon did not have this odd behavior, life would be very different (or maybe not even possible). Figure 2.9 shows bonding in carbon atoms.
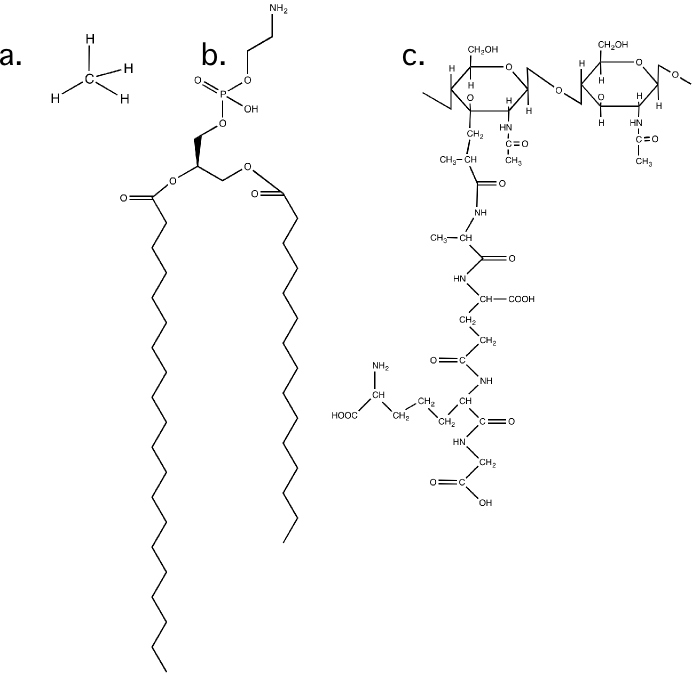
Figure 2.9. The arrangement of bonds in a carbon molecule. a) The structure of methane. Note how each bond is equidistant from the others b) A larger carbon molecule, in this case a membrane lipid. c) The structure of of a peptidoglycan molecule. These units will be put together to form part of the bacterial cell wall.
The names of carbon groups
As we examine the prokaryotes, certain chemical assemblies involving carbon will pop up over and over. Scientists have developed terms for these groups, and it is useful to learn this jargon to facilitate discussions about how the molecules of life behave and are put together. There are only a few of them, and it is worth spending the time to commit them to memory. These structures have important chemistries in biological molecules that help the larger structures in the cell perform their functions.
Table 2-4. Carbon groups often found in organic structures
| Group Name | Group Structure | Comments |
| Alcohol or Hydroxyl | 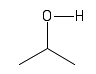 |
Hydroxyl groups decorate many different molecules and are important in chemical reactions and in holding larger molecules together. |
| Keytone | 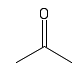 |
Carbon double bonded to oxygen. Important in reactions and structure. Often found in sugars |
| Aldehyde | 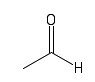 |
A keytone at the end of a molecule. These are also often found in sugars |
| Carboxyl | 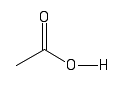 |
Under normal cell conditions, these groups will often lose there hydrogen. They are acidic and have a negative charge. |
| Amino | 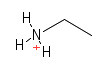 |
Nitrogen containing groups. They are found in proteins, DNA and many other structure in the cell. They will often pick up an extra hydrogen from the environment and give their containing molecule a positive charge. |
| Methyl | 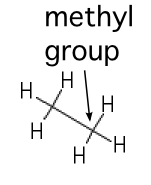 |
A carbon saturated with hydrogens. These groups tend to repel water |
| alkyl | Long chains of carbons tend to repel water and are useful for ordering structures in the cell. |
Stereochemistry is important for biological systems
In biological systems, stereoisomers are significant, especially in the case of sugars and amino acids. In many cases, organisms and their enzymes will only work with one type of molecule and not the other in much the same way as a glove only fits on one hand. Sugars that have identical molecular formulas but are mirror images of one another have been given the designations D and L to distinguish them. Biological systems, for the most part, prefer the D form of sugars. Similarly, amino acids can exist in either the D or L form, but in this case, life prefers the L form of amino acids (except for glycine, which does not have stereoisomers) (Figure 2.10). Why was one form chosen over the other? It was most likely a random event of evolution - when proteins were first evolving, L amino acids happened to be used to construct them, and this bias has been passed down through the ages. There are a few examples of the use of D amino acids for specific functions in living systems, for example, as part of the cell wall of bacteria.

Figure 2.10. The two isomers of alanine.. The L form of alanine is used in proteins, while the D form only finds use in the cell wall of bacteria.
We should note that a more modern system has been developed for naming stereoisomers. This system happens to use R (rectus) and S (sinister) as designations, but more importantly establishes a consistent method for naming any isomeric compound. The details are not important, but the student will see both D/L and R/S forms when referring to these types of geometries. Unfortunately for historical continuity, the D and L designations are used for amino acids and sugars and complicate things a bit when learning.
Key Takeaways
- A chemical formula and a structural formula are two ways to represent molecules. A chemical formula tells you the identity and number of each element, while a structural formula indicates how the elements are arranged in the molecule.
- Measuring and weighing molecules uses a system based upon the natural mass of atoms. Molecule weights are expressed as their molecular weight, or gram molecular weight. The amounts of molecules are expressed in moles, and their concentrations in solutions are expressed as molar.
- Water is the universal solvent for biological systems.
- The concentration of hydrogen ions, the pH, greatly influences biological chemistry and cells work hard to maintain an appropriate pH
- Carbon has the useful ability to rearrange its s and p orbitals, making it capable of forming four bonds instead of two. This allows the chaining together of an infinite number of carbons in unique arrangements, enabling the construction of life.
- Life has a handedness to it. D forms of sugars and L forms of amino acids are mostly used in the construction of biological molecules.
Quickcheck 2-3
Warning, you must be logged in to be able to have your exam graded. Answer the questions below and if you are a registered user of the site you will see a Grade Exam button. Click it to have your exam graded.